 Precipitating Sodium Chloride from its Solution. This can occur when solutions containing ionic compounds are mixed and an insoluble product is formed. How are precipitates used in everyday life?Ī precipitate is a solid formed in a chemical reaction that is different from either of the reactants. Whether or not such a reaction occurs can be determined by using the solubility rules for common ionic solids. Precipitation reactions occur when cations and anions in aqueous solution combine to form an insoluble ionic solid called a precipitate. Formation of kidney stone: The reaction between calcium ions and oxalic acid that are present in food results in calcium oxalate, which is a type of kidney stone. Precipitation Reaction Examples in Real Life Magnesium and calcium oxides are generally found in water pipes as precipitates. The rusting of unprotected iron in the presence of air and water is then inevitable because it is driven by an electrochemical process. Rust is then quickly produced by the oxidation of the precipitate. What is a precipitation reaction in chemistry? When a salt such as sodium chloride (table salt) dissolves in water, its ionic lattice is pulled apart so that the individual sodium and chloride ions go into solution. If an insoluble salt forms by the reaction of soluble substances in water and falls out of solution, we call it a precipitate (see image). 
What is an everyday example of a precipitate? AgNO3(aqueous) + NaCl(aqueous) - AgCl↓ + NaNO3 (aqueous). 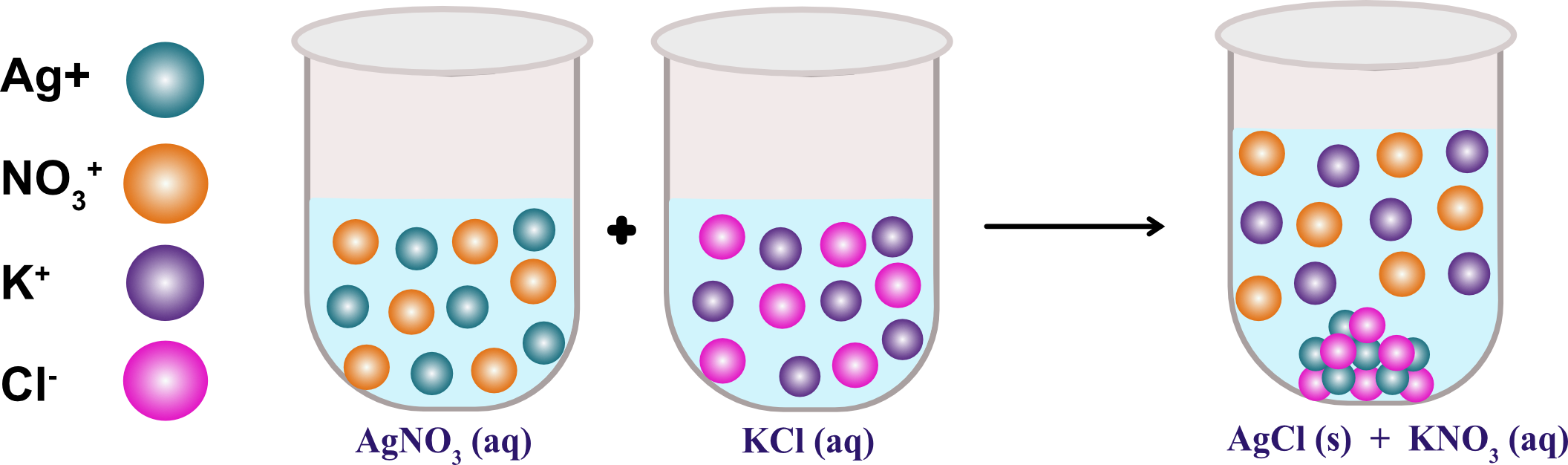
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
